Reactivation of HHV-6 determined by Coppe Labs’ immuohistochemical staining
Waukesha, WI, The November issue of the Journal of Clinical Virology (J Clin Virol. 2015 Nov; 72:82-4. doi: 10.1016/j.jcv.2015.09.013. Epub 2015 Oct 9) reports the first fatal case of biopsy-proven HHV-6 myocarditis post stem cell transplant. Dr. David Baewer, Chief Medical Officer at Coppe Laboratories co-authored the paper with colleagues from Sydney, Australia.
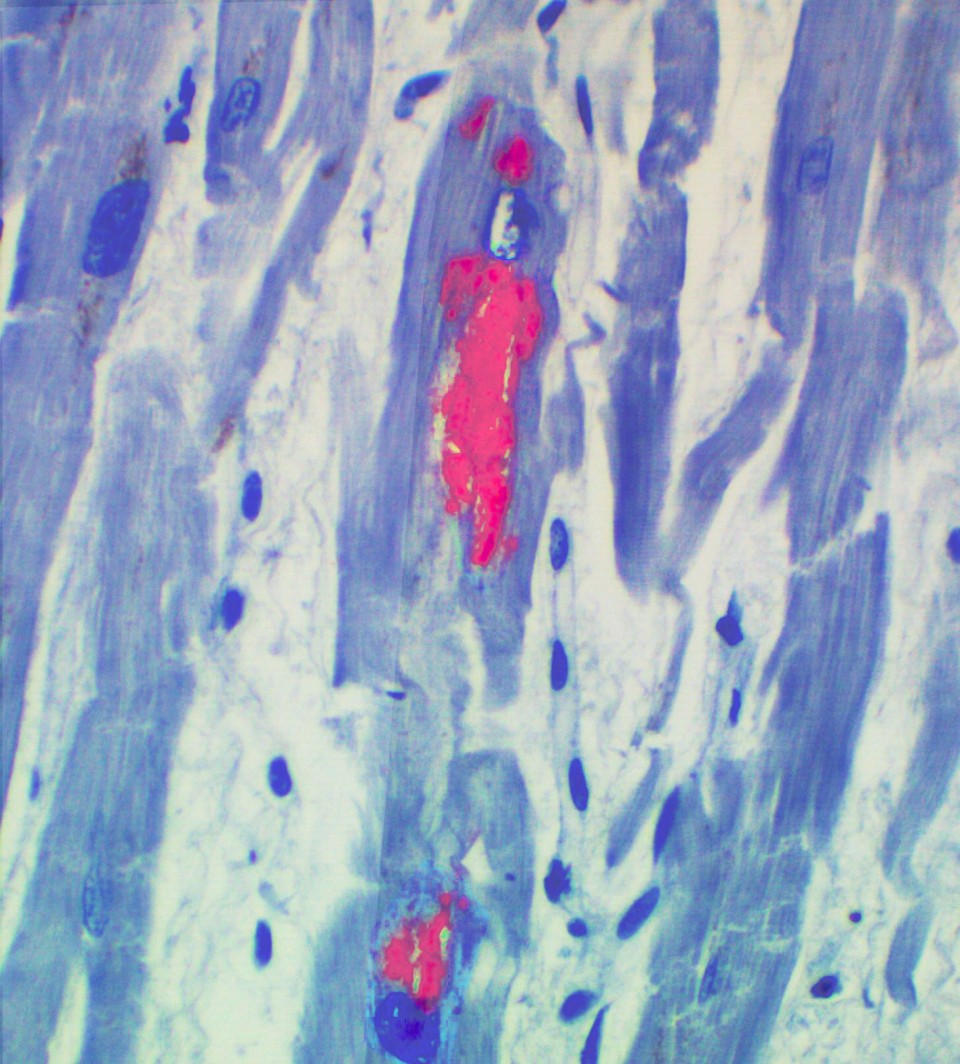
HHV-6 in cardiac myocytes
The publication reports the case of a 62-year old patient who underwent a matched, unrelated hematopoietic stem cell transplant. On day +13 the patient experienced cardiac failure. Treatment with diuretics, analgesic, and IV antibiotics failed to improve cardiac function. Despite growth factors and platelet transfusions the patient remained pancytopenic. HHV-6 DNA PCR was positive on peripheral blood with a low copy number (<200 copies/mL). Dilated cardiomyopathy and focal changes consistent with viral myocarditis were noted on post-mortem exam. Coppe Laboratories performed immunohistochemical staining on the cardiac tissue. Results indicated HHV-6 antigen in the cardiomyocytes.
This is the first publication of HHV-6 immunohistochemistry showing active HHV-6 infection in a heart biopsy of a stem cell transplant patient with fatal myocarditis. “HHV-6 is an opportunistic pathogen that can reactivate and cause a range of clinical symptoms in immunocompromised individuals,” said Dr. David Baewer, Chief Medical Officer of Coppe Laboratories. HHV-6 is treatable with antiviral therapy. This case illustrates the importance of recognizing the spectrum of organ damage that can result from reactivation of HHV-6 and the utility of immunohistochemistry staining to determine active HHV-6 infection. The picture (left) shows HHV-6 (red inclusions) in the cardiac myocytes.